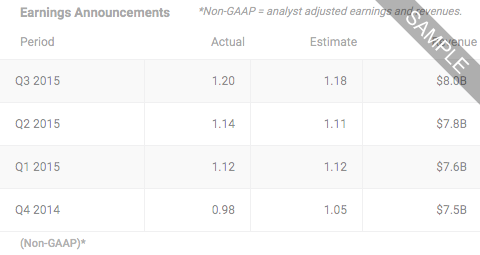
Applied Therapeutics IncNASDAQ:APLT
- LAST PRICE8.3700
- TODAY'S CHANGE (%)-0.0400 (-0.4756%)
- Bid / Lots8.3600/ 3
- Ask / Lots8.3800/ 2
- Open / Previous Close8.4800 / 8.4100
- Day Range
- 52 Week Range
- Volume248,987
Search Criteria
Filter search criteria using below inputs
Click on magnifying glass icon to search
| Company | Country | Symbol |
|---|
- TSX Comp
- TSX Venture
- DJIA
- S&P 500
- NASDAQ

| Time | Volume | APLT |
|---|---|---|
| 09:32 ET | 15219 | 8.5 |
| 09:34 ET | 1224 | 8.38 |
| 09:36 ET | 1852 | 8.355 |
| 09:38 ET | 5370 | 8.34 |
| 09:39 ET | 1000 | 8.41 |
| 09:41 ET | 8947 | 8.39 |
| 09:43 ET | 204 | 8.39 |
| 09:45 ET | 5304 | 8.48 |
| 09:48 ET | 7616 | 8.51 |
| 09:50 ET | 2300 | 8.49 |
| 09:52 ET | 1700 | 8.45 |
| 09:54 ET | 3161 | 8.46 |
| 09:56 ET | 8287 | 8.53 |
| 09:57 ET | 2222 | 8.475 |
| 09:59 ET | 1029 | 8.5 |
| 10:01 ET | 3304 | 8.505 |
| 10:03 ET | 22661 | 8.54 |
| 10:06 ET | 621 | 8.515 |
| 10:08 ET | 3673 | 8.48 |
| 10:10 ET | 27754 | 8.45 |
| 10:12 ET | 2558 | 8.47 |
| 10:14 ET | 4070 | 8.45 |
| 10:15 ET | 100 | 8.43 |
| 10:17 ET | 4428 | 8.47 |
| 10:19 ET | 1000 | 8.42 |
| 10:21 ET | 1679 | 8.41 |
| 10:24 ET | 4479 | 8.43 |
| 10:26 ET | 3750 | 8.465 |
| 10:28 ET | 13795 | 8.4 |
| 10:30 ET | 1348 | 8.37 |
| 10:32 ET | 8777 | 8.36 |
| 10:33 ET | 500 | 8.37 |
| 10:35 ET | 1299 | 8.375 |
| 10:37 ET | 1846 | 8.36 |
| 10:39 ET | 6496 | 8.41 |
| 10:42 ET | 4200 | 8.4 |
| 10:44 ET | 276 | 8.39 |
| 10:46 ET | 6418 | 8.36 |
| 10:48 ET | 400 | 8.35 |
| 10:50 ET | 3495 | 8.37 |
Sep 19, 2024
DJ Applied Therapeutics Is Maintained at Buy by Citigroup, from 11:40AM ET on Thursday Sep 19, 2024 by Dow Jones
11:40AM ET on Thursday Sep 19, 2024 by Dow JonesDJ Applied Therapeutics Is Maintained at Outperform by Leerink Partners, from 9:12AM ET on Thursday Sep 19, 2024 by Dow Jones
9:12AM ET on Thursday Sep 19, 2024 by Dow Jones
Sep 18, 2024
DJ Applied Therapeutics Up Over 67%, on Track for Record Percent Increase -- Data Talk, from 11:27AM ET on Wednesday Sep 18, 2024 by Dow Jones
11:27AM ET on Wednesday Sep 18, 2024 by Dow JonesDJ Applied Therapeutics Shares Rise as Drug Continues with Priority Review, from 7:59AM ET on Wednesday Sep 18, 2024 by Dow Jones
7:59AM ET on Wednesday Sep 18, 2024 by Dow JonesApplied Therapeutics Provides Regulatory Update on Govorestat for the Treatment of Classic Galactosemia, from 7:00AM ET on Wednesday Sep 18, 2024 by Dow Jones
7:00AM ET on Wednesday Sep 18, 2024 by Dow Jones
| Company sortable | Market Cap sortable | P/E Ratio (TTM) sortable | EPS Growth (5yr) sortable |
|---|---|---|---|
Applied Therapeutics Inc | 965.9M | -5.5x | --- |
AnaptysBio Inc | 981.3M | -5.1x | --- |
IGM Biosciences Inc | 940.8M | -4.6x | --- |
Sana Biotechnology Inc | 949.9M | -3.9x | --- |
Autolus Therapeutics PLC | 926.0M | -3.1x | --- |
Cullinan Therapeutics Inc | 1.0B | -5.6x | --- |
Company Information
Applied Therapeutics, Inc. is a clinical-stage biopharmaceutical company. It is focused on developing a pipeline of novel drug candidates against validated molecular targets in indications of high unmet medical need. The Company’s lead drug candidate, govorestat, is a novel central nervous system penetrant Aldose Reductase Inhibitor (ARI) for the treatment of central nervous system (CNS) rare metabolic diseases, including Galactosemia, Sorbitol Dehydrogenase (SORD) Deficiency, and PMM2-CDG. The Company is also developing AT-001, a novel potent ARI, for the treatment of Diabetic Cardiomyopathy (DbCM) a fatal fibrosis of the heart. It has completed a Phase I/II clinical trial evaluating AT-001 in approximately 120 patients with type 2 diabetes, in which no drug-related adverse effects or tolerability issues were observed. The preclinical pipeline also includes AT-003, an ARI designed to cross through the back of the eye when dosed orally, for the treatment of Diabetic retinopathy.
Contact Information
- Headquarters
- 545 FIFTH AVENUE, SUITE 1400NEW YORK, NY, United States 10017
- Phone
- 212-220-9226
- Fax
- 302-636-5454
Executives
- Chairman of the Board, President, Chief Executive Officer, Founder
- Shoshana Shendelman
- Chief Financial Officer and Principal Financial Officer, Director
- Leslie Funtleyder
- Chief Operating Officer and Chief Business Officer
- Constantine Chinoporos
- Interim Principal Financial Officer, Chief Accounting Officer
- Catherine Thorpe
- Chief Commercial Officer
- Dale Hooks
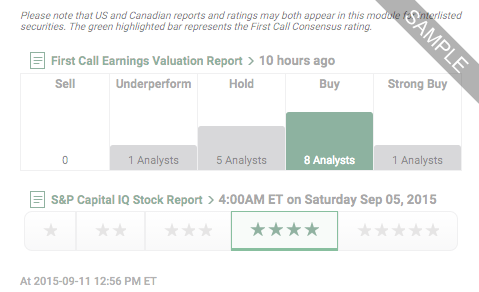
Our Ratings feature offers company-specific research ratings from providers such as First Call, S&P and Argus.
Open a New Account, or Login if you're a client.
You have access to a comprehensive selection of independent research reports from providers such as TD Securities, S&P, INK, and Argus.
Open a New Account, or Login if you're a client.
Market Cap | $965.9M |
|---|---|
Revenue (TTM) | $-333.0K |
Shares Outstanding | 114.8M |
Dividend Yield | 0.00% |
Annual Dividend Rate | --- |
Ex-Dividend Date | 01-01-01 |
Pay Date | 01-01-01 |
Beta | 2.04 |
EPS | $-1.52 |
Book Value | $-0.20 |
P/E Ratio | -5.5x |
Price/Sales (TTM) | --- |
Price/Cash Flow (TTM) | --- |
Operating Margin | --- |
Our Calendar feature allows you to view a wide selection of market and company events, including earnings, dividends, splits, rating changes, guidance, and more. Access past, current and future events in WebBroker.
Open a New Account, or Login if you're a client.