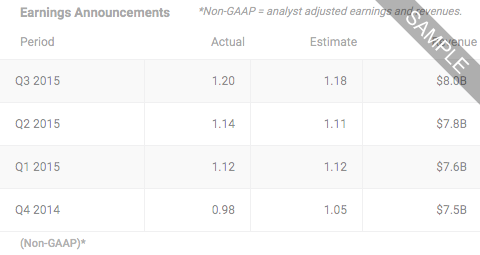
Cara Therapeutics IncNASDAQ:CARANASDAQ FSI:D. Deficient (Issuer failed to meet continued listing requirements)
- LAST PRICE0.6800
- TODAY'S CHANGE (%)0.0088 (1.3111%)
- Bid / Lots0.6800/ 32
- Ask / Lots0.6929/ 4
- Open / Previous Close0.6895 / 0.6712
- Day Range
- 52 Week Range
- Volume75,843
Search Criteria
Filter search criteria using below inputs
Click on magnifying glass icon to search
| Company | Country | Symbol |
|---|
- TSX Comp
- TSX Venture
- DJIA
- S&P 500
- NASDAQ

| Time | Volume | CARA |
|---|---|---|
| 09:32 ET | 12018 | 0.6802 |
| 09:34 ET | 100 | 0.68 |
| 09:36 ET | 960 | 0.6869 |
| 09:38 ET | 1396 | 0.68 |
| 09:39 ET | 5100 | 0.6893 |
| 09:41 ET | 5182 | 0.685 |
| 09:43 ET | 100 | 0.68 |
| 09:45 ET | 1024 | 0.68 |
| 09:48 ET | 559 | 0.68 |
| 09:50 ET | 100 | 0.68 |
| 09:52 ET | 1810 | 0.693 |
| 09:54 ET | 100 | 0.68 |
| 09:56 ET | 100 | 0.68 |
| 09:59 ET | 100 | 0.68 |
| 10:01 ET | 3084 | 0.6935 |
| 10:03 ET | 873 | 0.69 |
| 10:06 ET | 1145 | 0.6868 |
| 10:08 ET | 200 | 0.68335 |
| 10:10 ET | 4526 | 0.68 |
| 10:14 ET | 300 | 0.6816 |
| 10:15 ET | 12568 | 0.6836 |
| 10:17 ET | 500 | 0.6917 |
| 10:21 ET | 200 | 0.6935 |
| 10:24 ET | 584 | 0.692692 |
| 10:26 ET | 100 | 0.6934 |
| 10:28 ET | 200 | 0.6934 |
| 10:30 ET | 100 | 0.6934 |
| 10:32 ET | 200 | 0.69 |
| 10:33 ET | 100 | 0.6934 |
| 10:37 ET | 200 | 0.6934 |
| 10:39 ET | 400 | 0.6934 |
| 10:42 ET | 100 | 0.69 |
| 10:46 ET | 1399 | 0.6934 |
| 10:48 ET | 300 | 0.69 |
| 10:53 ET | 200 | 0.6934 |
| 10:55 ET | 100 | 0.6917 |
| 10:57 ET | 784 | 0.69 |
| 11:00 ET | 100 | 0.6934 |
| 11:04 ET | 7041 | 0.6866 |
| 11:09 ET | 435 | 0.6831 |
| 11:11 ET | 300 | 0.684 |
| 11:13 ET | 3500 | 0.68 |
| 11:18 ET | 300 | 0.68 |
| 11:20 ET | 400 | 0.6929 |
| 11:26 ET | 300 | 0.68 |
| 11:29 ET | 400 | 0.68 |
| 11:31 ET | 100 | 0.68 |
May 23, 2024
DJ Cara Therapeutics Price Target Maintained With a $5.00/Share by Needham, from 6:37AM ET on Thursday May 23, 2024 by Dow Jones
6:37AM ET on Thursday May 23, 2024 by Dow Jones
May 15, 2024
DJ Cara Therapeutics Price Target Maintained With a $3.60/Share by HC Wainwright & Co., from 11:47AM ET on Wednesday May 15, 2024 by Dow Jones
11:47AM ET on Wednesday May 15, 2024 by Dow Jones
May 14, 2024
DJ Cara Therapeutics Price Target Maintained With a $5.00/Share by Needham, from 11:16AM ET on Tuesday May 14, 2024 by Dow Jones
11:16AM ET on Tuesday May 14, 2024 by Dow Jones
May 13, 2024
*DJ Cara Therapeutics 1Q Loss/Shr 56c >CARA, from 4:01PM ET on Monday May 13, 2024 by Dow Jones
4:01PM ET on Monday May 13, 2024 by Dow JonesCara Therapeutics Reports First Quarter 2024 Financial Results, from 4:01PM ET on Monday May 13, 2024 by Dow Jones
4:01PM ET on Monday May 13, 2024 by Dow Jones
| Company sortable | Market Cap sortable | P/E Ratio (TTM) sortable | EPS Growth (5yr) sortable |
|---|---|---|---|
Cara Therapeutics Inc | 36.7M | -0.3x | --- |
ImmuCell Corp | 35.4M | -8.9x | --- |
Aadi Bioscience Inc | 45.2M | -0.7x | --- |
Turnstone Biologics Corp | 60.1M | -0.8x | --- |
Prenetics Global Ltd | 70.4M | -1.3x | --- |
SQZ Biotechnologies Co | 752.0K | 0.0x | --- |
Company Information
Cara Therapeutics, Inc. is a development-stage biopharmaceutical company. The Company is focused on treatment paradigm to improve the lives of patients suffering from chronic pruritus. It is developing an oral formulation of difelikefalin, a selective, predominantly peripherally acting, non-scheduled Kappa opioid receptor agonist, for the treatment of chronic neuropathic pruritus associated with Notalgia Paresthetica (NP), a common, underdiagnosed neuropathy affecting the upper back. It is conducting Phase II/III Clinical Program in NP. The program is comprised of two studies - KOURAGE 1 and KOURAGE 2, which are double-blind, placebo-controlled, eight-week studies with patients allowed to roll-over into open-label 52-week extensions. It has developed an IV formulation of difelikefalin, for the treatment of moderate-to-severe pruritus associated with advanced chronic kidney disease in adults undergoing hemodialysis. The IV formulation is out-licensed worldwide.
Contact Information
- Headquarters
- 400 Atlantic Street, Suite 500STAMFORD, CT, United States 06901
- Phone
- 203-406-3700
- Fax
- 203-567-1510
Executives
- Independent Chairman of the Board
- Martin Vogelbaum
- President, Chief Executive Officer, Director
- Christopher Posner
- Chief Financial Officer
- Ryan Maynard
- Chief Compliance Officer, General Counsel, Secretary
- Scott Terrillion
- Director
- Helen Boudreau
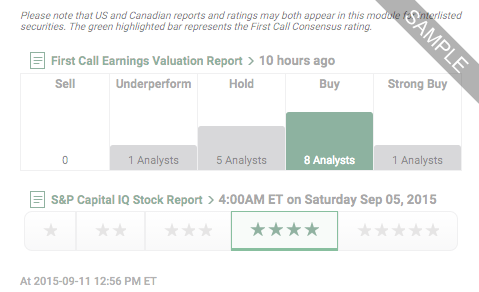
Our Ratings feature offers company-specific research ratings from providers such as First Call, S&P and Argus.
Open a New Account, or Login if you're a client.
You have access to a comprehensive selection of independent research reports from providers such as TD Securities, S&P, INK, and Argus.
Open a New Account, or Login if you're a client.
Market Cap | $36.7M |
|---|---|
Revenue (TTM) | $16.9M |
Shares Outstanding | 54.7M |
Dividend Yield | 0.00% |
Annual Dividend Rate | --- |
Ex-Dividend Date | 01-01-01 |
Pay Date | 01-01-01 |
Beta | 0.79 |
EPS | $-2.26 |
Book Value | $1.05 |
P/E Ratio | -0.3x |
Price/Sales (TTM) | 2.2 |
Price/Cash Flow (TTM) | --- |
Operating Margin | -729.19% |
Our Calendar feature allows you to view a wide selection of market and company events, including earnings, dividends, splits, rating changes, guidance, and more. Access past, current and future events in WebBroker.
Open a New Account, or Login if you're a client.