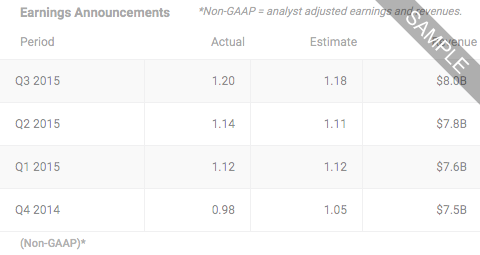
Xenon Pharmaceuticals IncNASDAQ:XENE
- LAST PRICE35.8700
- TODAY'S CHANGE (%)-1.1300 (-3.0541%)
- Bid / Lots35.8500/ 4
- Ask / Lots36.0700/ 4
- Open / Previous Close36.8400 / 37.0000
- Day Range
- 52 Week Range
- Volume84,192
Search Criteria
Filter search criteria using below inputs
Click on magnifying glass icon to search
| Company | Country | Symbol |
|---|
- TSX Comp
- TSX Venture
- DJIA
- S&P 500
- NASDAQ

| Time | Volume | XENE |
|---|---|---|
| 09:32 ET | 12837 | 36.415 |
| 09:34 ET | 1500 | 36.3 |
| 09:36 ET | 700 | 36.6 |
| 09:38 ET | 700 | 36.57 |
| 09:39 ET | 5100 | 36.59 |
| 09:41 ET | 300 | 36.61 |
| 09:45 ET | 400 | 36.04 |
| 09:48 ET | 7000 | 36.37 |
| 09:50 ET | 100 | 36.11 |
| 09:54 ET | 200 | 36.4 |
| 09:57 ET | 15042 | 36.54 |
| 09:59 ET | 1507 | 36.31 |
| 10:01 ET | 2118 | 36.25 |
| 10:03 ET | 300 | 36.19 |
| 10:06 ET | 600 | 36.26 |
| 10:08 ET | 950 | 36.2575 |
| 10:10 ET | 100 | 36.2 |
| 10:12 ET | 100 | 36.355 |
| 10:14 ET | 600 | 36.3575 |
| 10:17 ET | 200 | 36.4 |
| 10:19 ET | 18320 | 36.27 |
| 10:21 ET | 200 | 36.29 |
| 10:24 ET | 514 | 36.19 |
| 10:26 ET | 200 | 36.19 |
| 10:28 ET | 100 | 36.33 |
| 10:30 ET | 200 | 36.23 |
| 10:32 ET | 704 | 36.31 |
| 10:33 ET | 200 | 36.22 |
| 10:35 ET | 100 | 36.04 |
| 10:37 ET | 100 | 36.01 |
| 10:39 ET | 200 | 36.095 |
| 10:42 ET | 200 | 35.96 |
| 10:44 ET | 100 | 35.83 |
| 10:46 ET | 100 | 35.87 |
Jun 18, 2024
DJ Xenon Pharmaceuticals Price Target Maintained With a $55.00/Share by RBC Capital, from 8:59AM ET on Tuesday Jun 18, 2024 by Dow Jones
8:59AM ET on Tuesday Jun 18, 2024 by Dow Jones
Jun 5, 2024
Xenon Pharmaceuticals to Present at the Goldman Sachs 45th Annual Global Healthcare Conference, from 4:01PM ET on Wednesday Jun 05, 2024 by Dow Jones
4:01PM ET on Wednesday Jun 05, 2024 by Dow Jones
May 28, 2024
| Company sortable | Market Cap sortable | P/E Ratio (TTM) sortable | EPS Growth (5yr) sortable |
|---|---|---|---|
Xenon Pharmaceuticals Inc | 2.8B | -13.2x | --- |
Geron Corp | 2.7B | -12.4x | --- |
SpringWorks Therapeutics Inc | 2.7B | -7.0x | --- |
IDEAYA Biosciences Inc | 2.7B | -17.7x | --- |
MoonLake Immunotherapeutics | 2.5B | -52.5x | --- |
Biohaven Ltd | 3.1B | -5.0x | --- |
Company Information
Xenon Pharmaceuticals Inc. is a Canada-based clinical-stage biopharmaceutical company focused on developing therapeutics to improve the lives of patients with neurological disorders, with a focus on epilepsy. The Company’s product pipeline includes XEN1101 (Focal Onset Seizures), XEN1101 (Primary Generalized Tonic-Clonic Seizures), XEN1101 (Major Depressive Disorder) and NBI-921352 (NEUROCRINE). It is developing XEN1101, a differentiated Kv7 potassium channel opener, for the treatment of epilepsy, MDD and potentially other neurological disorders. Its XEN1101 Phase III epilepsy program includes two identical Phase III clinical trials, called X-TOLE2 and X-TOLE3, that are designed closely after the Phase IIb X-TOLE clinical trial. Its Phase III X-ACKT clinical trial is intended to support potential regulatory submissions in an additional epilepsy indication of primary generalized tonic-clonic seizures (PGTCS).
Contact Information
- Headquarters
- 3650 Gilmore WayVANCOUVER, BC, Canada V5G 4W8
- Phone
- 604-484-3300
- Fax
- 604-484-3450
Executives
- Chairman of the Board
- Simon Pimstone
- President, Chief Executive Officer, Director
- Ian Mortimer
- Chief Financial Officer
- Sherry Aulin
- Executive Vice President - Drug Discovery
- James Empfield
- Executive Vice President - Strategy and Innovation
- Robin Sherrington
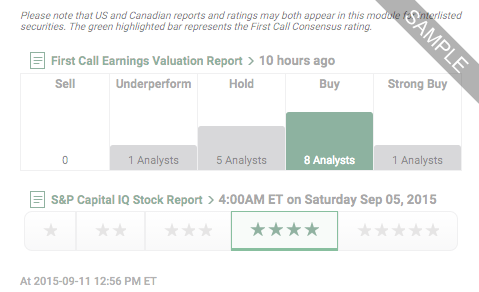
Our Ratings feature offers company-specific research ratings from providers such as First Call, S&P and Argus.
Open a New Account, or Login if you're a client.
You have access to a comprehensive selection of independent research reports from providers such as TD Securities, S&P, INK, and Argus.
Open a New Account, or Login if you're a client.
Market Cap | $2.8B |
|---|---|
Revenue (TTM) | $0.00 |
Shares Outstanding | 75.5M |
Dividend Yield | 0.00% |
Annual Dividend Rate | --- |
Ex-Dividend Date | 01-01-01 |
Pay Date | 01-01-01 |
Beta | 1.26 |
EPS | $-2.71 |
Book Value | $12.31 |
P/E Ratio | -13.2x |
Price/Sales (TTM) | --- |
Price/Cash Flow (TTM) | --- |
Operating Margin | --- |
Our Calendar feature allows you to view a wide selection of market and company events, including earnings, dividends, splits, rating changes, guidance, and more. Access past, current and future events in WebBroker.
Open a New Account, or Login if you're a client.