GH Research PLCNASDAQ:GHRS
- LAST PRICE8.0000
- TODAY'S CHANGE (%)0.2600 (3.3592%)
- Bid / Lots7.1300/ 2
- Ask / Lots12.0000/ 1
- Open / Previous Close7.7700 / 7.7400
- Day Range
- 52 Week Range
- Volume72,289
Search Criteria
Filter search criteria using below inputs
Click on magnifying glass icon to search
| Company | Country | Symbol |
|---|
- TSX Comp
- TSX Venture
- DJIA
- S&P 500
- NASDAQ

| Time | Volume | GHRS |
|---|---|---|
| 09:32 ET | 734 | 7.77 |
| 09:34 ET | 440 | 7.75 |
| 09:54 ET | 100 | 7.99 |
| 10:10 ET | 3000 | 7.79 |
| 10:14 ET | 100 | 7.9 |
| 10:15 ET | 200 | 7.65 |
| 10:17 ET | 100 | 7.65 |
| 10:21 ET | 300 | 7.725 |
| 10:26 ET | 500 | 7.68 |
| 10:28 ET | 100 | 7.68 |
| 10:44 ET | 188 | 7.3409 |
| 10:50 ET | 100 | 7.66 |
| 10:53 ET | 102 | 7.5932 |
| 10:55 ET | 285 | 7.4168 |
| 11:02 ET | 400 | 7.64 |
| 11:09 ET | 100 | 7.68 |
| 11:11 ET | 100 | 7.64 |
| 11:26 ET | 100 | 7.77 |
| 11:38 ET | 206 | 7.539 |
| 12:43 ET | 100 | 7.55 |
| 12:45 ET | 100 | 7.5967 |
| 12:59 ET | 100 | 7.58 |
| 01:32 ET | 200 | 7.59 |
| 01:51 ET | 5211 | 7.77 |
| 02:24 ET | 250 | 7.694 |
| 02:27 ET | 100 | 7.87 |
| 02:36 ET | 500 | 7.77 |
| 02:38 ET | 398 | 7.7 |
| 02:40 ET | 1800 | 7.665 |
| 02:49 ET | 1421 | 7.725 |
| 02:51 ET | 1506 | 7.7 |
| 03:20 ET | 14300 | 7.76 |
| 03:23 ET | 650 | 7.77 |
| 03:27 ET | 200 | 7.85 |
| 03:32 ET | 100 | 7.85 |
| 03:38 ET | 100 | 7.88 |
| 03:41 ET | 1785 | 7.9 |
| 03:43 ET | 100 | 7.9 |
| 03:45 ET | 100 | 7.94 |
| 03:48 ET | 1501 | 7.94 |
| 03:50 ET | 200 | 7.95 |
| 03:52 ET | 1100 | 7.96 |
| 03:54 ET | 7100 | 7.82 |
| 03:56 ET | 900 | 7.9 |
| 03:57 ET | 300 | 7.9 |
| 03:59 ET | 1137 | 8 |
| Company sortable | Market Cap sortable | P/E Ratio (TTM) sortable | EPS Growth (5yr) sortable |
|---|---|---|---|
GH Research PLC | 402.7M | 0.0x | --- |
Alto Neuroscience Inc | 389.6M | --- | --- |
Biomea Fusion Inc | 416.8M | -2.9x | --- |
enGene Holdings Inc | 379.4M | -2.0x | --- |
Compass Pathways PLC | 429.5M | -2.7x | --- |
Nautilus Biotechnology Inc | 371.2M | -5.2x | --- |
Company Information
GH Research PLC is a clinical-stage biopharmaceutical company. The Company is engaged in offering treatment for psychiatric and neurological disorders. The Company's pipeline includes GH001, GH002 and GH003. The Company focuses on developing its 5- Methoxy-N, N-Dimethyltryptamine, (5-MeO-DMT), therapies for the treatment of patients with Treatment Resistant Depression (TRD). Its product candidate, GH001, is formulated for 5-MeO-DMT administration via an inhalation approach. With GH001, the Company has completed two Phase 1 healthy volunteer clinical trials and a Phase 1/2 clinical trial in patients with treatment-resistant depression (TRD). The GH002 is a 5-MeO-DMT product candidate formulated for administration via an injectable approach. GH002 has completed pre-clinical development to support early clinical trials. The GH003 is a 5-MeO-DMT product candidate formulated for administration via an intranasal administration approach.
Contact Information
- Headquarters
- Joshua Dawson House, Dawson StreetDublin, Ireland D02 RY95
- Phone
- ---
- Fax
- ---
Executives
- Non-Executive Chairman of the Board
- Florian Schonharting
- Chief Executive Officer
- Velichka Valcheva
- Non-Executive Independent Vice Chairman of the Board
- Michael Forer
- Vice President - Finance
- Julie Ryan
- Managing Director, Ireland
- Magnus Halle
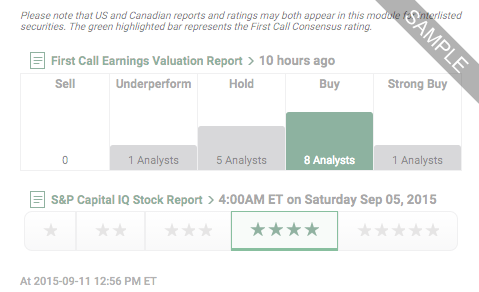
Our Ratings feature offers company-specific research ratings from providers such as First Call, S&P and Argus.
Open a New Account, or Login if you're a client.
You have access to a comprehensive selection of independent research reports from providers such as TD Securities, S&P, INK, and Argus.
Open a New Account, or Login if you're a client.
Market Cap | $402.7M |
|---|---|
Revenue (TTM) | --- |
Shares Outstanding | 52.0M |
Dividend Yield | 0.00% |
Annual Dividend Rate | --- |
Ex-Dividend Date | 01-01-01 |
Pay Date | 01-01-01 |
Beta | 0.80 |
EPS | --- |
Book Value | $4.21 |
P/E Ratio | 0.0x |
Price/Sales (TTM) | --- |
Price/Cash Flow (TTM) | --- |
Operating Margin | --- |
Our Calendar feature allows you to view a wide selection of market and company events, including earnings, dividends, splits, rating changes, guidance, and more. Access past, current and future events in WebBroker.
Open a New Account, or Login if you're a client.